Precision control in biomedical research is critical for the success of any experiment. This is especially true for gas mixing and blending applications. These gas mixing applications often involve the creation of artificial atmospheres or other gas mixtures. To get statistically sound results, scientists must achieve precision flow control in creating these mixtures for sensitive experiments. In practice, two or more gases are mixed (blended) in a chamber until the specified concentrations (or partial pressures) of each gas are achieved. Flow controllers are commonly used to create these gas mixtures.
Precision flow control of multiple gases at low flows
Achieving stable flow control in gas mixing and blending applications can be challenging because of the difficulty in mixing unique gases and the difficulty of controlling lower flows. During calibration, most flow controllers are "curve-fit" for a particular gas because they are not linear devices. Due to this, in most gas mixing applications, scientists need to use multiple mass flow controllers — each one calibrated for one of the component gases. Another challenge is precision control at low flows. Since the chamber is filled with the component gases, the pressure will increase, which will result in backpressure at the measuring instrument. Some flow controllers, especially those based on differential pressure (ΔP), will lose accuracy as backpressure changes. Differential pressure devices also perform poorly with low flows because they require a pressure drop to operate. At low flows, of course, a low pressure drop occurs, so the sensitivity of the meter may not be sufficient.
That brings up the obvious consideration: What type of flow control technology should be used in research? The two common methods for measuring and controlling flow rates are mass flow and volumetric flow. In most research, especially those involving biological or chemical reactions, mass is the quantity of interest. Mass flow depends on the number of molecules in the flow, not the volume occupied by these molecules. This volume will change with temperature or pressure. Higher pressure means the same number of molecules must occupy a smaller volume (assuming the temperature stays constant). In contrast, when temperatures are higher, the volume occupied by these molecules expands (assuming the pressure stays constant). These two facts are based on the Ideal Gas Law.
The Ideal Gas Law
PV = nRT
Where:
P is the pressure of the gas
V is the volume of the gas
n is the amount of substance of gas (in moles)
R is the ideal gas constant
T is the absolute temperature of the gas
Mass flow controllers that use thermal mass flow technology are ideal for biomedical research because they provide scientists with direct mass flow control, which is unaffected by temperature and pressure fluctuations.
Suspended animation research uses mass flow controllers at low flows
Like many biomedical scientists, Dr. David Kraus, a sulfide biologist at the University of Alabama, had a difficult gas mixing and blending application for his research on suspended animation. Dr. Kraus’ experiments required gas mixing of two gases, strictly specified gas compositions and precision control at low flows.
Imagine being suspended in time — metabolism slows, heart rate decreases and body temperature drops. Dr. Kraus and other scientists are discovering that, with small amounts of hydrogen sulfide (H2S), scientists can buy time in suspended animation. The key to the success of these experiments is to use mass flow controllers to deliver precise and repeatable amounts of H2S to laboratory mice — too much H2S will kill the mice, while the right amount induces suspended animation. This suspended animation state reduces their metabolism and heart rates, virtually freezing their bodies in time. With the removal of the H2S, the mice "come back to life."
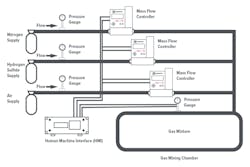
Most controllers are used for common gases (air, argon and nitrogen). However, Dr. Kraus’ research uses a special blend of 5 percent hydrogen sulfide and 95 percent nitrogen with a very low flow range of 1 to 10 standard cubic centimeters per minute (sccm) mixed into another stream of combined air and nitrogen mixture (see Figure 2).
Figure 2. Typical gas mixing setup with mass flow controllers
Dr. Kraus had some concerns about the ability to accurately measure mixed gases at these low flows. Because of the known benefits of capillary thermal mass flow technology, he chose a mass flow controller for his research with dial-a-gas capability. Dr. Kraus used the on-board dial-a-gas capability to program a custom listing for his 5 percent H22S blend. Then in the laboratory, Dr. Kraus could select this programmed mixture from the dial-a-gas menu or use the same instrument to measure any of the other nine preprogrammed gases.
Dr. Kraus’ concerns about accuracy are well-founded. With traditional mass flow controllers, gas mixtures are notorious for inaccuracy. During calibration, most instruments are curve-fit to the application because they are not linear. They are calibrated with a primary gas, but a gas mixture has different properties than the primary gas, so the original curve is not correct for the mixture. However, the mass flow controller Dr. Kraus used has advanced laminar flow technology, which creates a linear relationship that allows the K-factors programmed into the flow controller to correct for any gas blend over the entire flow range. This enables the dial-a-gas capability discussed above, so now identical controllers can control the various pure gases that make up the mixture and deliver the mixture, all without recalibration. This type of linearity means the mass flow controller is only calibrated at zero and full scale (see Figure 1).
In addition, the powerful solenoid valve of the mass flow controller provides precision control and accuracy even at the very low flow range of 1 to 10 sccm. This low of a flow rate is difficult to obtain with differential pressure devices since low flows do not generate much pressure drop to begin with. Because of the inaccuracies at low flow, using differential pressure devices for this experiment could have resulted in the delivery of too much hydrogen sulfide. Instead, the experiment was a success. No mice died from the toxic H2S gas and all the subjects reached suspended animation.
Figure 1. Thermal mass flow controllers are ideal for precision control at low flows.
Benefits of capillary thermal mass flow controllers
Through his biomedical research, Dr. Kraus observed a suspended animation-like state in his laboratory mice. Their heart rates decreased from 650 to 50 beats per minute and breathing rates dropped from 140 to 20 breaths per minute. Their metabolic rates and core body temperatures also decreased, suspending their bodies in time. And when the H2S was removed, the mice returned to normal.
According to Dr. Kraus, these suspended animation breakthroughs will not only change the face of the medical field, but now NASA plans to use suspended animation for cryosleep chambers so astronauts can hibernate while traveling long distances in space. In 2007, Dr. Mark Roth received a MacArthur Genius Award for his work with H2S-induced suspended animation using mass flow controllers.
Only with precision mass flow controllers that provide stable, smooth and accurate flow control of multiple gases
at low flow rates would Dr. Kraus been able to conduct these sensitive experiments with such success. Flow control instrumentation can actually help change the world.
Scott A. Rouse serves as vice president of product management at Sierra Instruments, where he has been employed for nearly two decades. Rouse obtained his bachelor’s degree in chemical engineering from the University of Texas at Austin. He can be reached at [email protected] or 800-866-0200. Learn more at sierrainstruments.com/smarttrak.