The process of disinfecting potable water has been evolving over time due to the implementation of more stringent health and safety requirements. Historically, potable water has been disinfected using free chlorine at a residual level of ~0.5 PPM total residual chlorine; however, chloramine treatment of potable water by municipalities is becoming more common at a residual level of ~4.0 PPM total residual Cl2. This change has proven to be detrimental to elastomeric seals used for both point-of-use and distribution-system applications. Choosing the correct sealing material will ensure that the seal will last within the predicted life of the system.
Chloramination of potable water is the process of creating chloramines for disinfection by combining chlorine and ammonia. Chloramination has become widespread due to more stringent water quality standards. These standards have been implemented to ensure that drinking water is safe for consumption. One of the criteria for safe drinking water is to limit the amount of disinfection by-products (DBPs) formed en route to the consumer. DBPs are carcinogenic and the levels have been regulated by the EPA since 1979. DBPs are formed when chlorine reacts with excess organic material in the water.
Water disinfection involves chlorine reacting with harmful substances in the water to kill all matter harmful to humans. However, chlorine-containing byproducts with detrimental health effects are formed during this process. These byproducts can include chloroform and carbon tetrachloride. The primary disinfection process involves disinfecting with free chlorine to kill all microorganisms. After this process has been completed, ammonia is added to form chloramines to retain residual disinfection while the water is being transported.
READ ALSO: Is a Double-Mechanical Seal Necessary?
Chloramination has been used as a secondary disinfection method for over 100 years. It is typically not used as the primary method since the effectiveness is much less than that of free chlorine for disinfection. Although chloramines do not have the kill power of free chlorine (roughly 20 percent less1), chloramines have a much longer residual life. Residual life is very important in potable water. Water may travel through several miles of pipe to reach the final destination. During this journey, organic matter has the potential to be introduced into the system. Since free chlorine can dissipate quickly, water that has traveled a long distance may not be as safe as intended. Chloramines ensure the water still has the ability to kill microorganisms further downstream, thus creating safer water. Chloramines also do not include the offensive taste and odor that accompanies potable water disinfected with free chlorine.
Chloramine Chemistry
Chloramine is formed when chlorine and ammonia combine in an aqueous environment. Primary disinfection of potable water can be accomplished using either chlorine or ammonia. Upon the completion of primary disinfection, the second component (chlorine or ammonia) is added to form chloramines. The most common method is to chlorinate the water prior to adding the ammonia. This guarantees adequate disinfection prior to adding the ammonia. Once the chlorine and ammonia are added, the reaction begins. The reaction products are determined by the rate of reaction. The three distinct molecules are monochloramine (NH2Cl), dichloramine (NHCl2), and nitrogen trichloride (NCl3). The desired primary molecule for disinfection is monochloramine. The rate at which each molecule is formed is dependent on two variables; pH of the water and ratio of reagents added (chlorine to ammonia). The desired reaction product is monochloramine for several reasons. Monochloramine is a better disinfectant than dichloramine and nitrogen trichloride. In addition, the taste and odor is offensive when dichloramine and nitrogen trichloride levels increase. The addition of chlorine and ammonia are critical to the formation of a primarily monochloramine solution. The basic reaction of chlorine and ammonia to form chloramines is shown in Figure 1.
| NH3(aq) + HOCl D NH2Cl + H2O (Monochloramine) NH2Cl + HOCl D NHCl2 + H2O (Dichloramine) NHCl2 + HOCl D NCl3 + H2 (Nitrogen Trichloride) Figure 1. Mechanism for forming chloramines1 |
As previously stated, the formation of a monochloramine solution is dependent on the pH of the water and the ratio of chlorine to ammonia. At a pH of 5.5 and greater, mostly monochloramine is produced with some dichloramine. Above a pH of seven, there is monochloramine and only a trace amount of dichloramine produced. Nitrogen trichloride is formed at a pH of less than three.
The second criterion for producing primarily monochloramine is the ratio of chlorine to ammonia. The optimum ratio for the formation of monochloramine is four mg chlorine/one mg ammonia as N (four-to-one). As the ratio of chlorine to ammonia is increased, dichloramine begins to form along with the monochloramine. At a ratio of chlorine to ammonia above eight-to-one, only dichloramine and nitrogen trichloride are produced.
Elastomer Comparison
The rubber industry has a variety of polymer types to choose from when a new sealing application arises Key questions needed to determine seal and material type are: minimum and maximum temperatures, fluids to seal, and if the seal will be static or dynamic. From this information a material recommendation is made. The common polymer types currently used in the potable water industry are listed below along with some of their characteristics.
Ethylene Propylene (EPM, EPDM): EPDM is currently the workhorse for the potable water industry due to the excellent resistance to water and especially chlorine-containing water. Seals utilizing EPDM rubber can be operated over a continuous temperature range of -40 C to 150 C, with the seals being capable of intermittent spikes up to 175 C. EPDM can be compounded to meet a wide variety of mechanical properties including durometer, tensile, elongation, and compression set. Compression-set resistance of EPDM seals is far superior to seals made from thermoplastic and thermoplastic vulcanizate (TPV) materials.
EPDM materials serve a wide variety of industries and provide excellent sealing ability for a range of fluids. However, this family of materials is not recommended for hydrocarbon applications (petroleum grease, fuel, etc.). When exposed to hydrocarbons, EPDM material exhibit severe swelling and rapid loss of mechanical properties.
Nitrile Butadiene (NBR): This elastomer is typically used in the potable water industry when there is a concern that the seal will come into contact with a hydrocarbon-based material. Nitrile rubber performs well in water, but has poor weatherability. The polymer backbone contains carbon-carbon double bonds (unsaturation), which are weaker and more susceptible to weather and ozone attack than a fully saturated polymer such as EPDM or silicone.
Silicone (VMQ): Silicone rubber is the best option when it comes to chloramine resistance. The drawbacks to silicone rubber are cost and mechanical properties. VMQ does not have the mechanical properties of other elastomer families. The poorer mechanical properties are abrasion resistance, tear strength, and tensile strength. However, silicone is exceptionally chloramine resistant and also moderately resistant to petroleum oils.
Analysis
Chloramine attack on rubber materials has been described in several ways. The typical methods of seal failure are loss of sealing force or gland overfills. When the failure mode is loss of sealing force, the rubber has typically been eroded away from the seal. This can lead to noticeable buildup in faucet screens or the water will visibly contain small pieces of the seal. Once surface degradation begins, it will progress quickly. The second type of failure, gland overfill, is a result of the seal swelling excessively. Excessive swell can rupture the mating components and cause a leak path. If the assembly does not rupture, there is a potential the water flow path will be closed off due to the swelled seal.
| Degradation Key 1. Complete degradation (entire surface), floaters 2. Complete degradation (entire surface), no floaters 3. Internal degradation – blisters 4. Severe swelling – degradation 5. Edge degradation – with floaters 6. Edge degradation (visible) or surface degradation 7. Severe swelling – no degradation 8. Edge degradation with slight surface degradation 9. Slight edge/surface degradation (no visual signs) 10. No degradation Table 1. Key for rating tested samples for degradation and swell (developed by Parker Hannifin’s Engineered Seals Division). |
The standard method for testing the chloramine resistance of rubber is defined in standard ASTM D6284. This standard gives the recipe for preparing the chloramine solution, as well as the concentration used for testing and specimen size. All testing and results performed by Parker Hannifin (www.parker.com) are per the below conditions.
- 50 ppm total residual chlorine
- Solution is changed daily
- (except weekends and holidays)
- Temperature is constant at 70 C
- Volume of solution to surface area of the
- rubber specimen is 20 mL/cm2 minimum
- Sample size is 1 x 2 x 0.040 inch unless otherwise stated
- Total residual chlorine is the combination of all chlorine-containing compounds in the water. This includes free chlorine, monochloramine, dichloramine, and nitrogen trichloride. Preparing the solution per ASTM D6284 makes certain that monochloramine is the prevailing chlorine compound.
A variety of materials were tested during the evaluation of chloramines-resistant materials. The test matrix included the base polymers EPDM (Ethylene Propylene), VMQ (Silicone), NBR (Nitrile), HNBR (Hydrogenated Nitrile), FKM (Fluorocarbon), and ECO (Epichlorohydrin).
Degradation Methods
When exposed to chloramine containing water, the method of degradation varies widely depending on the base polymer and the formulation. The main modes of degradation are severe swelling, edges swelling with degradation, degradation without swell, and crazing (surface attack). Each type of degradation poses unique sealing problems. A key to determine the type and severity of degradation was developed by Parker Hannifin’s Engineered Seals Division to aid in the analysis of the samples. The key in Table 1 ranks the characteristics of the material on a scale of one to 10 (10 = best).
Examples of each type are shown below:
Elastomers for Sealing
The degree of chloramine resistance is not the only criterion evaluated for sealing applications. In order for a seal to perform, the seal must exert enough force on the mating components to form the seal. As seals are aged in the compressed state, the opportunity arises for the seals to permanently deform and take the new shape of the compressed state. This is known as compression set.
Compression set is tested in the laboratory by compressing a sample a known amount, aging the sample (usually elevated temperature), releasing the force from the sample and allowing the sample to cool. If the sample does not return to the original height, it has taken a set. Samples are measured for initial and final height and the percentage of set is calculated. Low compression set is desired for seals. 100 percent set indicates that when compressed, the seal permanently deforms to its compressed height. This result is undesired as it indicates that the seal will rapidly lose sealing force over time.
Compression set measured in the chloramine solution is useful to determine the effect of the fluid on the seal. The fixture (Figure 6) used for testing is a non-corrosive fixture that allows solution to contact both the OD and ID of a sample. O-ring samples or a sample die-cut from an ASTM slab of uniform thickness can be used for the test. Examination of the samples after exposure shows the samples are only degraded where the solution is contacting the seal (ID and OD). The top and bottom of the sample are unaffected. Figure 7 shows the degradation only where the solution is in contact with the seal.
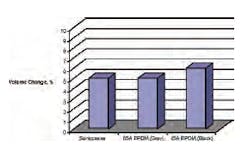
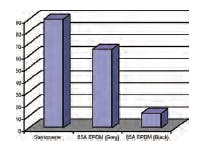
Looking at Figure 8, the volume changes for all three materials are very similar. Figure 9 is a comparison of the compression-set properties and depicts a dramatic difference between materials.
After 1008 hours of aging in the chloramine solution, the Santoprene material has taken a set of >85 percent in the chloramine fluid. As shown, similar formulations of EPDM materials can provide very different compression-set properties. The 85A grey material has a much higher compression set than the black, >60 percent and <10 percent, respectively.
Chloramine-Resistant Materials
The acceptance criterion for chloramines-resistant materials is zero degradation and minimal swelling after aging at the specified conditions.
Parker Hannifin, for example, has developed several new chloramines-resistant materials. Once a chloramines-resistant material was developed (zero degradation, minimal swell), these materials were tested against a competitive material, also marketed as chloramines-resistant.
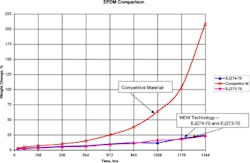
A second type of test specimen was used for the chloramine testing in addition to the standard specimen. The dimensions are 1.47 in ID, 1.75 in OD, and a height of 0.137 in. Figure 10 depicts the weight change against time (in weeks). The sample type is listed below each chart in the description. Figure 10. Chloramine testing on rings (1.47” x 1.75” x 0.137”), competitive material, Rating = 1 vs. Parker’s EJ273-70: Rating = 9, and EJ274-70: Rating = 9.
The EPDM comparison in Figure 11 depicts a significant reduction in swell as well as degradation. The competitive material is completely degraded and severely swelled (>200%) after eight weeks.As shown in Figures 11, Parker Hannifin’s new technology EPDM materials show improved property retention compared to the competitive and standard materials.
Parker Hannifin has two material offerings for chloramine resistance; EJ273-70 Figure 11. Parker’s EJ274-70 on the left and the competitive material on the right after chloramine testing. EJ274-70 Rating = 9, competitive material = 1.
and EJ274-70 (internally lubricated). These materials are universal compounds and will be used to make engineered shapes, complex over-molded seals, lathe-cut seals or o-rings. Along with its molding capability, Parker Hannifin offers design engineering assistance and material development opportunities for your specific application. The design assistance includes seal and mating component design recommendations, as well as FEA analysis. All compounds are available for sampling.
Valerie Combs is a senior chemical engineer and laboratory manager at Parker Hannifin’s Engineered Seals Division. For the past four years Ms. Combs has been instrumental in the areas of compound development for the industrial, military, aerospace, and transportation markets. Ms. Combs’s expertise has provided several solutions to a variety of industry problems in these markets. Ms. Combs earned a bachelors degree in Chemical Engineering from Tri-State University in Angola, Indiana and is an active member of SAE, ACS-Rubber Division, and the Fort Wayne Rubber Group.
References
- American Water Works Association, Research Foundation (AWAARF), 1993, Chloramine Effects on Distribution System Materials, Denver, CO.: AWWARF.
- Keller, Robert C. Ed. Krishna C. Baranwal and Howard L. Stephens, Basic Elastomer Technology, 1st Ed., Baltimore, Md., The Rubber Division, American Chemical Society.